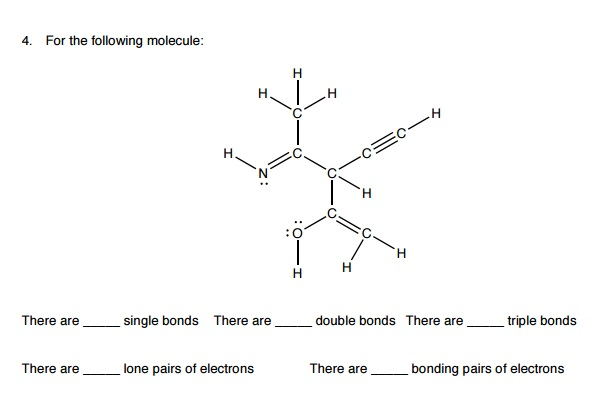
Which of the following species has a trigonal bypyramid structure? a. The bond angles about the carbon atoms in the formaldehyde molecule, H2C=O, are about:ģ8. How many of the following molecules or ions are linear? NH3 NH4+ HCN CO2 NO2 a. Trigonal bipyramidal square pyramidal distorted tetrahedral octahedral none of theseģ6. III, V I, III, IV II, IV, V I, III, IV, VI none of them These molecules have a zero net dipole moment. I, II, IV I, III, IV, VI III, V, VI I, IV, VI I, II, IV, VIģ4. SF4 Select the molecule(s) that fit the given statement. Use the following to answer questions 33-34: Consider the following molecules. Which carbon in this molecule has tetrahedral bonding? a. How many nonbonding electrons appear in the Lewis structure of this molecule? a. How many electrons must be shown in the Lewis structure of this molecule? a. Use the following to answer questions 29-31: Consider the compound crotonaldehyde, whose skeleton is HĢ9. In the Lewis structure for elemental nitrogen there is (are) a. (Hint: Consider formal charges.) a)Ĭ) d) e) 28. Choose the electron dot formula that most accurately describes the bonding in CS2. How many resonance structures can be drawn for the molecule O3? a. How many of the following molecules – SF2, SF4, SF6, SiO2- are polar? a. In the cyanide ion (CN-), the nitrogen has a formal charge of a. N S P I All of the atoms (a-d) can exceed the octet rule.Ģ4.
Which of the following atoms cannot exceed the octet rule in a molecule? a. different or the same, depending on the conditions leading to maximum repulsion. the same because in each case there are the same number of electron pairs around the central atom. the same because both nitrogen and carbon are both in the second period. different because in each case there are a different number of electron pairs around the central atom. different because in each case there are a different number of atoms around the central atom.

According to the VSEPR model, the arrangement of electron pairs around NH3 and CH4 are a. O3 OF2 HCN H2O More than one of the above have equally large bond angles.Ģ2.

Of the following, which molecule has the largest bond angle? a. For the elements Rb, F, and O, the order of increasing electronegativity is: a. Atoms with very similar electronegativity values are expected to form a. Linear molecules cannot have a net dipole moment.Ģ. A molecule with very polar bonds can be nonpolar. The electrons in a polar bond are found nearer to the more electronegative element. Dipole moments result from the unequal distribution of electrons in a molecule. Ionic bonding results from the transfer of electrons from one atom to another. Which of the following statements is incorrect? a. AP Chemistry Practice Test #6 Chapter 8 and 9 1.
